Dr. Koji Watari (Leader) and Dr. Kimiyasu Sato (Research Scientist) of the Advanced Sintering Technology Group, the Advanced Manufacturing Research Institute (Director: Hideto Mitome) of the National Institute of Advanced Industrial Science and Technology (AIST) (President: Hiroyuki Yoshikawa) have discovered the conditions for uniform dispersion of boron nitride (BN) filler in organic solvents and plastics. These conditions facilitated the fabrication of a composite material of filler (dispersed particles) and plastic, and a high-thermal-conductivity composite plastic film containing dispersed inorganic particles was successfully prepared.
Today's smaller, more powerful electronic devices, communications equipment, and lighting apparatus require optimum solutions for heat dissipation, prompting the development of plastic materials with high heat dissipation effect. The thermal conductivity of plastics has generally been enhanced by adding powder fillers such as inorganic oxides. Inorganic nitrides have higher thermal conductivity than oxides and could potentially improve the heat dissipation effect; however, it has been very difficult to achieve uniform dispersion of nitride fillers in plastics. In this research, the use of BN filler having many functional groups on its surface and high compatibility with organic solvents and plastics made it easy to disperse the filler in polymer (plastic) varnish and improve the thermal conductivity of a inorganic particle dispersed plastic composite film.
The results will be presented at a workshop hosted by the Advanced Manufacturing Research Institute of AIST, under the title "Latest trends and research and development in engineering plastic materials – hybridization with inorganic materials," to be held on October 31, 2008 in Tokyo.
 |
|
High-thermal-conductivity composite plastic film with dispersed BN fillers |
Plastic wiring boards and packages that efficiently dissipate generated heat are in great demand due to the higher integration of electronic circuits into smaller, denser semiconductor packages resulting from rapid development in electronics and communications fields. For next-generation portable information devices, there are high expectations for high-thermal-conductivity films in terms of portability. LED lighting products must use very powerful for highly brightness. However, in this case, the conventional plastic cannot dissipate the heat from the LEDs efficiently.
Plastic materials having high formability and a high degree of design flexibility are also used in automobile-related fields. Today's smaller, more powerful products require countermeasures against heat generation that was not previously regarded as a problem, and demand for materials with high thermal conductivity is growing.
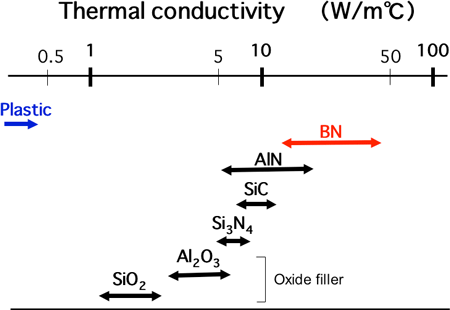
Thermal conductivity of general-purpose plastics ranges from 0.1 to 0.5 W/m°C. Efforts have already been made to improve the thermal conductivity of plastics by adding oxide fillers such as alumina and silica. Thermal conductivity of the oxide fillers is, however, low and ranges between 1 and 6 W/m°C, as shown in Figure 1. Thus, thermal conductivity of plastic composites containing dispersed oxide fillers remains at only 1 to 3 W/m°C. On the other hand, the thermal conductivity of fillers of boron nitride, silicon carbide, and aluminum nitride is 5 to 40 times as high as that of oxide fillers. However, they have poor dispersive properties in organic solvents due to the smaller number of functional groups on their surfaces. In addition, they have low compatibility with plastics. Accordingly, it has been difficult to achieve uniform dispersion of these fillers in plastics.
 |
|
Figure 1. Thermal conductivity of inorganic fillers. The values were obtained by fabrication of green pellets with filler and subsequent thermal conductivity measurement. |
The Advanced Sintering Technology Group, the Advanced Manufacturing Research Institute of AIST, has been engaged in research and development of dispersion techniques for inorganic particles, evaluation techniques for surface states of inorganic materials, and surface treatment techniques. As part of this work, the group focused on boron nitride, which has high thermal conductivity.
The addition of inorganic fillers (particles) is an effective method for enhancing the thermal conductivity of plastics. Inorganic filler provides an advantage if it meets the following conditions:
-
Shows high thermal conductivity
-
Consists of fine particles
-
Has functional groups on the particle surface for improved compatibility with organic solvents and plastics
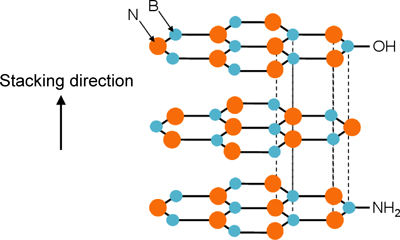
Boron nitride (BN), which shows relatively high thermal conductivity among inorganic materials (see Fig. 1), is used as the filler in this study. The surfaces of BN fine particles have functional groups including -OH (hydroxyl group) and -NH2 (amino group). The groups mainly bond covalently to the BN crystal side faces, which are perpendicular to stacking planes of a hexagonal network structure with alternately bonded boron and nitrogen atoms (see Fig. 2). These functional groups allow the BN filler to easily disperse in organic solvents. Moreover, the existence of -OH allows the filler and plastics to bond together by means of a coupling agent. However, if the particles become coarse due to excessive growth of BN crystals, uniform dispersion in plastics becomes difficult.
 |
|
Figure 2. Crystal structure of BN (hexagonal) and function groups bonding covalently to the BN crystal side faces. |
 |
|
Figure 3. Scanning electron microscope image of used BN fillers |
In this research, we evaluated various kinds of BN particles as candidate fillers and then selected the one that showed superior dispersive properties in organic solvents. Figure 3 shows an electron microscope image of the filler, which consists of fine particles of nearly uniform size, and an average particle size of about 0.8 μm. These fine BN particles show restricted crystal growth on the plane perpendicular to the stacking planes, and the side-face area is larger than that of the stacking planes.
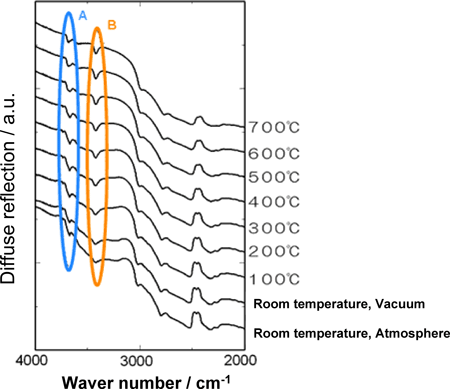
Figure 4 shows infrared spectra of the BN filler, recorded by diffuse reflection measurement. A series of absorption bands (peaks projecting downward) in the range circled and indicated by the letter 'A' represents the absorption band of -OH covalently bonded to boron atoms. Similarly, a series of absorption bands in the range circled and indicated by the letter 'B' represents the absorption band of -NH2 covalently bonded to boron atoms. Many of these functional groups exist on the side faces of the crystals. Both absorption bands were observed at high temperature and under reduced pressure, which means that -OH and -NH2 groups are stable in such conditions. These stable functional groups contribute to enhanced compatibility of the filler with organic solvents and plastics.
 |
|
Figure 4. Spectra of diffuse-reflection infrared spectroscopy of BN filler |
Figure 5 (a) shows a schematic diagram of the BN filler used in this research, and Fig. 5 (b) shows a schematic diagram of the other BN fillers available commercially.
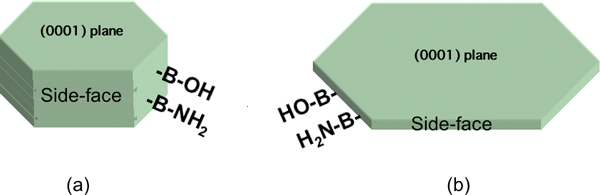 |
Figure 5. Schematic diagram of BN fillers.
(a) used one (b) commercial available one. |
Infrared spectra of the other commercial BN fillers measured using diffuse reflection mentod revealed that the intensity of absorption bands of -OH and -NH2 covalently bonded to boron atoms is low and, therefore, the number of functional groups is very small. The reason for this is as follows: In ordinary BN particle synthesis, the (0001) plane grows faster than the side-face and area of the side-face decreases relatively. Therefore, BN filler such as that shown in Figure 5 (b) has a smaller number of functional groups as a whole and has poor dispersive properties in organic solvents. Poor dispersion results in air remaining in the gaps between the aggregated fillers, leading to low thermal conductivity.
We prepared a plastic film containing dispersed inorganic particles by adding the selected BN filler to a polyamide acid varnish and then mixing and hot-pressing it to form a film of about 100 μm in thickness. Figure 6 shows the dependence of the thermal conductivity of the polyamide film on the amount of filler added. Thermal conductivity was improved as the amount of filler was increased, and it reached as high as 7 W/m°C when the amount of added filler was 60%.
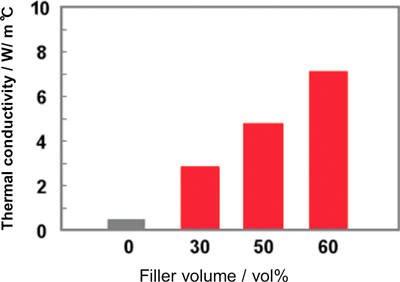 |
|
Figure 6. Relationship between thermal conductivity of composite plastic film with dispersed BN fillers and filler volume |
Based on these findings, we intend to investigate the hybridization of various fillers and organic materials and the further improvement of thermal conductivity.